- Home
- Weddings
- Portraits
- Journal
- Contact
- Night vision scope mountrd on filr
- Top off dj khaled mp3 download
- How to add developer tab on microsoft word 2008 for mac
- Tong hua li piano
- Ps4 mass effect andromeda download
- Dune 2000 download full game free
- Stellar phoenix photo recovery mac crack
- External hard drive not mounting mac paragon
- Homo lumo organic chemistry
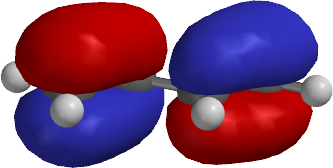
The energy gap is determined by the balance between the through space interaction and the through bond interaction. The ground state spin-multiplicity, singlet (S) versus triplet (T), of the 1,3-diradicals CP1 and CB1 will be determined by the energy gap between the nonbonded molecular orbitals (NBMO) ψ S and ψ A that are occupied by the two electrons in the 1,3-diyl structures (Figure 4 ). How can one control the ground state spin-multiplicity? Therefore, at least the singlet ground state is needed to have the character of C–π–C bonds in the cyclic structures. 11 The π-single bonding character between the two carbons should be possible only in the singlet state. However, the ground state spin-multiplicities for CP1a and CB1a (X = H) are known to be triplets. 10 To observe such highly reactive species and characterize their structure and features, one may lower the temperature using liquid nitrogen or helium. 10 The energy difference between CP1a and the bicyclic σ-bonding compound RC1a was determined to be ca. 9 The energy barrier of the σ-bond formation in CP1a (X = H) was reported to be very small, i.e. The two candidate molecules are known to be intermediates for the thermal isomerization of the corresponding bicyclic compounds. Therefore, cyclopentane-1,3-diyl ( CP1a) and cyclobutane-1,3-diyl ( CB1a) structures are promising candidates for molecules with π-single bonding (Figure 3 ). 27, 54, and 64 kcal mol −1, 8 respectively. The strain energy of cyclopropane, bicyclopentane ( RC1a), and bicyclobutane ( RC2a) systems is known to be ca. Molecular design for π-single bonded species. 6 Although it is easy to draw such an “unusual” bonding system by hand or using computer drawing programs, an experimental study to demonstrate such bonding would be highly challenging and involve new areas of chemistry.įigure 3. 5 In addition to the potential for the unique electronic character, the molecular structure would be quite unusual, owing to the resulting planar four-coordinate carbons. If such bonding is possible, the resulting extremely small HOMO–LUMO energy gaps are expected to provide molecules with new features, including altered optical properties and redox activity. In this Highlight Review article, our study of the literature since 1998 is summarized, with the aim of answering the naive question “Is π-single carbon–carbon bonding (C–π–C) possible?” (Figures 1 and 2 ). 3 In metal–metal bonding systems, the iron atoms in Fe 2(CO) 6 are known to be connected only by π-bonding of the metal d orbitals. In 2013, Fischer, Frenking, and their co-workers reported the Ge–Ge π single bonding (Ge–π–Ge) in Ga 2Ge 2 molecules (Figure 1 ). 2 Although the molecule shows considerable diradical reactivity, it should possess π-single bonding character between the boron–boron atoms (B–π–B) owing to the most stable electronic configuration of the singlet state. In 2002, Bertrand and co-workers isolated the B 2P 2 singlet diradicaloid species (Figure 1 ). π-Single bonded species (C–π–C) and related molecules, B 2P 2 and Ge 2Ga 2. In 2012, however, the bonding characteristics of C 2 were reinvestigated by Shaik and co-workers, and revised to a quadruple bond interaction between the two carbon atoms, which includes two π-bonds, one inner σ-bond, and one outer σ-bond. Is it possible to connect carbon atoms with only π-bonding and without σ-bonds, C–π–C (Figure 1 )? The diatomic molecule C 2 had been believed to be a 1,2-dicarbene species possessing two π-bonds without σ-bonds, i.e. the highest-occupied-molecular-orbital (HOMO) and the lowest-unoccupied-molecular-orbital (LUMO), are generally in the reactive bonding system in π-conjugated molecules. In most cases the π-electron systems play crucial roles in molecular function, because the frontier molecular orbitals, i.e. Therefore, as in the ethylene molecule (CH 2=CH 2), π-bonding is possible in conjunction with underlying σ-bonding. The σ-bonds are more energetically stable than the π-bonds. σ- and π-Bonds are possible for carbon–carbon connections in organic molecules. There are four well-known concepts in chemical bonding: σ-, π-, δ-, and φ-bonding. These structural features originate from chemical bonds. The properties of molecules, which include reactivity, light-absorption, light-emission, and redox properties, are controlled by their electronic and three-dimensional structures.
